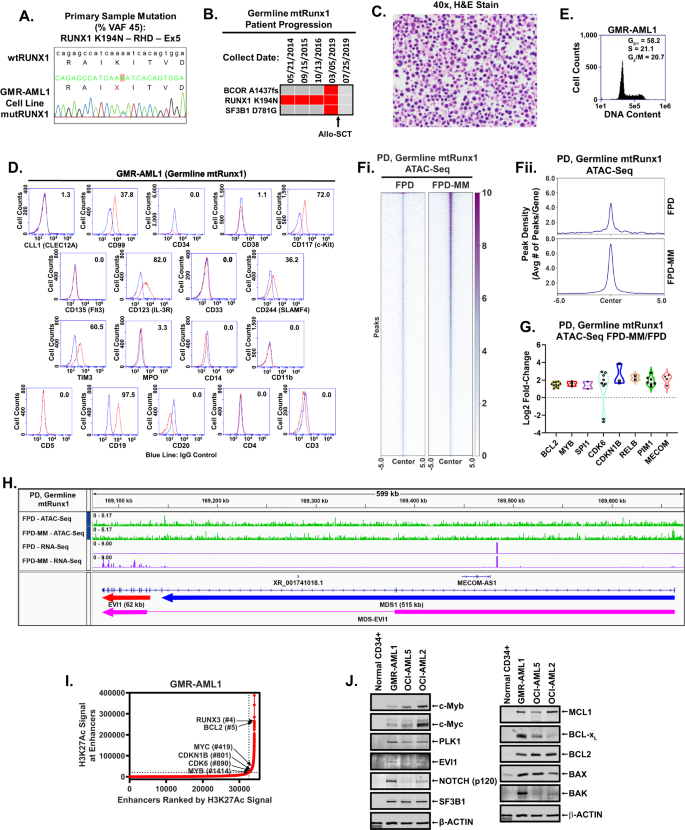
Establishing the primary FPD-MM GMR-AML cell line
To handle the necessity to develop related in vitro and in vivo fashions and take a look at novel focused therapies for FPD-MM, we efficiently established the primary, repeatedly cultured cell line (GMR-AML1) expressing germline mtRUNX1. This cell line was generated from the BMA cells from a affected person with FPD-MM that developed from RUNX1-FPD expressing mtRUNX1 K194N (Fig. 1A). On this affected person, development to FPD-MM was related to co-mutations, together with BCOR A1437fs (VAF 13%) and SF3B1 D781G (VAF 4%), as documented by NGS (Fig. 1B). Notably, the mutations recognized by NGS within the FPD-MM cells from the affected person weren’t detected by NGS within the GMR-AML1 cell line aside from the RUNX1 K194N, which was additionally recognized in different pedigree members who had developed FPD (Fig. S1A). Moreover, one of many members of the pedigree additionally exhibited transformation of FPD to FPD-MM. This attests to the biologic significance of the presence of the germline RUNX1 K194N mutation. GMR-AML1 cells have been cytogenetically diploid and lacked MYC or MLL1 rearrangement, or different copy quantity positive factors or losses on array CGH (Fig. S1B, C and never proven). As a substitute, entire exome sequencing (WES) carried out on GMR-AML1 cell line recognized extra mutations in TP53 (P72R), AIM2 (K340fs), NELFB (L523F), CEP152 (Y370X), SUGP2 (H23L), RRM2B (R71fs), TADA3 (T27R), SPDYE6 (G292C) and PRDM9 (S814R) with % VAF ranging between 33 to 55% (Fig. S1D). The practical significance of TP53 codon 72 alteration for the transformation to FPD-MM is unclear [18]. Collectively, these genetic alterations counsel that GMR-AML1 cell line is derived from the clonal growth beneath in vitro tradition circumstances of AML stem-progenitor cells current within the BMA of the affected person with FPD-MM. The in vitro survival and development of GMR-AML1 cells is probably going promoted by these WES-detected mutations. We subsequent additional characterised the biologic options of GMR-AML1 cell line expressing mtRUNX1. Determine 1C exhibits the morphologic options of the GMR-AML1 cells. Most of those cells are poorly differentiated myeloid progenitors. As decided by move cytometry, GMR-AML1 cells categorical CD117, CD123, CD99, CD33, TIM3, CD86, and CD18, however not CD34, CLEC12A, CD38, CD244, CD11b, CD14, CD3 and CD5 (Fig. 1D). Circulate cytometry additionally confirmed the cell cycle distribution of GMR-AML1 cells, with 58.2, 21.1 and 20.7% of cells in G1, S and G2/M section of the cell cycle, respectively (Fig. 1E). Moreover, tail vein-infused GMR-AML1 cells in NSG mice engrafted within the mice, leading to AML development and mortality of all mice in 4 to five weeks put up engraftment (vide infra, Fig. 2E).
A Electropherogram of the Runx1 mutation recognized in GMR-AML1. B Oncoplot of mutations recognized by Subsequent-Era Sequencing (NGS) throughout development of a germline mutant Runx1 affected person. C H & E stained cells from a consultant Formalin-Mounted Paraffin-Embedded (FFPE) part exhibiting the morphology of GMR-AML1 cells. D Circulate cytometry evaluation of AML related myeloid, progenitor, and B/T cell markers. E GMR-AML1 cells in exponential section have been harvested and stuck for cell cycle evaluation. Panel exhibits consultant DNA content material/cell cycle standing as decided by move cytometry. F World ATAC-Seq warmth map and peak profile in longitudinal FPD-MM vs. FPD at peak +/− 5 kb decision. G The log2 fold-increase (>1.25-fold) in accessibility of genes in FPD-MM vs. FPD as recognized by ATAC-Seq evaluation. H IGV plot exhibiting peak density of Tn5-accessible chromatin and RNA-Seq peak profile in major, patient-derived FPD-MM and FPD samples. I GMR-AML1 cells in exponential section have been crosslinked with formaldehyde. H3K27Ac ChIP-Seq evaluation was carried out on sonicated chromatin. Ranked ordering of tremendous enhancers (ROSE) evaluation was carried out. Rank of particular person tremendous enhancers are proven relative to the entire variety of tremendous enhancers. J Baseline protein expressions in GMR-AML1 cells in comparison with regular HPCs or leukemia cell strains OCI-AML5 (somatic mtRunx1) and OCI-AML2 (wtRunx1).
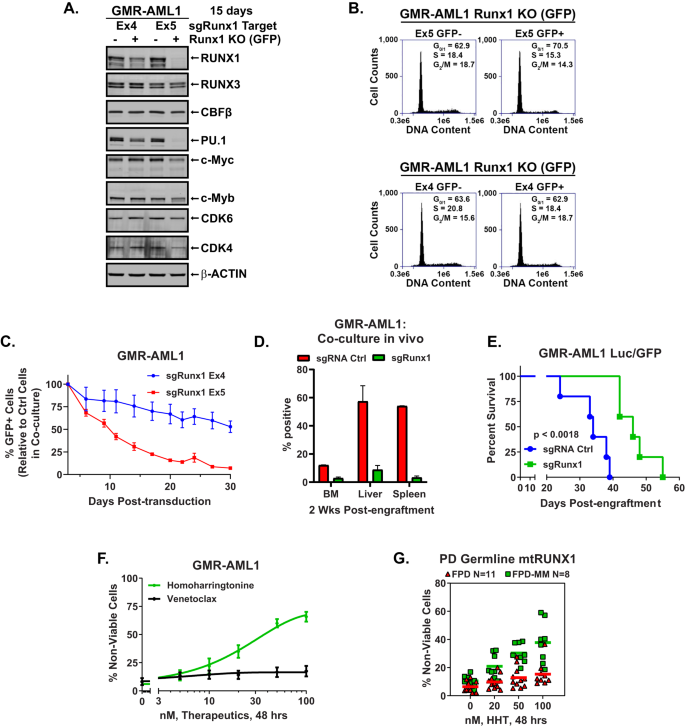
A GMR-AML1/Cas9 cells have been transduced with lentivirus expressing Runx1 information RNAs and eGFP for 72 h. Cells have been sorted by FACS into GFP optimistic and detrimental populations and cultured for an extra 12 days. Following this immunoblot analyses have been carried out on complete cell lysates. The expression of β-Actin within the lysates served because the loading management. B GMR-AML1/Cas9 cells transduced and cultured as in (A). have been harvested and stuck for cell cycle evaluation. Panel exhibits consultant DNA content material/cell cycle standing as decided by move cytometry. C GMR-AML1/Cas9 cells transduced as in (A). have been blended (+/− GFP) and co-cultured for 30 days. The % GFP+ cells have been measured each 3–4 days by move cytometry. The road represents the imply % of GFP+ cells from 4 experiments +/− S.E.M. D GMR-AML1/Cas9 cells transduced as in (A). with sgRNA Ctrl (tdTomato) or sgRunx1 Ex5 (eGFP) have been cultured for seven days then blended at a 1:1 ratio and tail vein injected into pre-irradiated NSG mice. Mice have been monitored day by day for 2 weeks after which two mice have been euthanized. Bone marrow, liver, and spleen have been harvested and analyzed for tdTomato and GFP optimistic cells by move cytometry. E GMR-AML1/Cas9 cells have been transduced and cultured as in (D). and individually injected into NSG mice (N = 5 per cohort). The Kaplan-Meier survival curve exhibits the median and general survival of NSG mice bearing GMR-AML1/Cas9 cells transduced with sgRNA management or sgRunx1. Significance was decided by a Mantel-Cox log rank take a look at. F GMR-AML1 cells have been handled with the indicated concentrations of HHT or venetoclax for 48 h. Then the proportion non-viable cells have been decided by TO-PRO-3 iodide staining and move cytometry. G PD germline mutant Runx1 FPD and FPD-MM cells have been handled ex vivo with the indicated concentrations of HHT for 48 h. Then the proportion non-viable cells have been decided by TO-PRO-3 iodide staining and move cytometry.
Alterations in chromatin accessibility and gene expressions in FPD-MM and GMR-AML1 cells
We subsequent carried out ATAC-Seq and RNA-Seq analyses on cells harvested longitudinally from a affected person on the RUNX1-FPD versus FPD-MM stage [19]. Determine 1F demonstrates that, in comparison with RUNX1-FPD, FPD-MM cells exhibited a major improve in chromatin accessibility. Notably, log2-fold improve in ATAC-Seq peaks was noticed at a number of loci, together with MECOM, PIM1, RELB, CDKN1B, CDK6, SPI1, MYB and BCL2 loci, whereas ATAC-Seq peaks declined on the PRDM9, WNT5A, TERT and AF3 loci (Figs. 1G and S1E). RNA-Seq evaluation confirmed vital improve in 404 mRNA expressions and decline in 1305 mRNA expressions in FPD-MM cells, as in comparison with RUNX1-FPD cells (Fig. S1F). QPCR evaluation confirmed vital improve in BCL2 and MYB however decline in RUNX1 and SPI1 mRNA expressions (Fig. S1G). Notably, ATAC-Seq and RNA-Seq peaks considerably elevated on the MECOM locus, concerned in AML stem cell renewal and differentiation arrest [20], in FPD-MM in comparison with RUNX1-FPD cells (Fig. 1H). We subsequent carried out ChIP evaluation with anti-H3K27Ac antibody in GMR-AML cells [4, 19]. This demonstrated excessive occupancy of H3K27Ac on the energetic super-enhancers of 1478 genes, together with these of MYC, RUNX3, MYB, CDKN1B, BCL2 and CDK6 (Fig. 1I) [21]. To evaluate gene expressions, we carried out immunoblot analyses to check protein expressions in GMR-AML1 cells with germline mtRUNX1 versus OCI-AML5 and OCIAML2 cells, which categorical somatic heterozygous mtRUNX1 and two copies of wtRUNX1 [12], respectively, in addition to versus CD34+ regular hematopoietic progenitor cells. Determine 1J demonstrates that, in comparison with regular CD34+ progenitor cells, GMR-AML1 cells expressed increased ranges of c-Myb, EVI1, c-Myc, polo-like kinase 1 (PLK1), NOTCH1, BCL2, BcL-xL, MCL1, BAX and BAK. Nevertheless, in comparison with OCI-AML5, GMR-AML1 cells expressed decrease ranges of c-Myb, c-Myc and BCL2 (Fig. 1J). These findings spotlight the elevated exercise of chromatin and elevated expression of particular oncoproteins which can be related to arrested differentiation, elevated in vitro development and in vivo leukemogenic potential of GMR-AML1 cells.
Impact of RUNX1 depletion on cell development and sensitivity to homoharringtonine (HHT, omacetaxine) in GMR-AML1 and FPD-MM cells
We subsequent decided the consequences of RUNX1 depletion by CRISPR knock out (KO) on cell development and drug sensitivity of GMR-AML1 cells. These cells, transduced with and expressing Cas9, have been additional transduced with a lentivirus expressing a RUNX1 gRNA (focusing on both exon 4 or 5) and EGFP. Forty-eight hours after transduction, the cells have been sorted into GFP optimistic (Runx1 KO) or detrimental management (Ctrl) populations. Immunoblot evaluation confirmed that, in comparison with the exon 4 gRNA, exon 5 gRNA-mediated RUNX1 KO led to better depletion of RUNX1, PU.1, c-Myc, c-Myb, CDK4/6, however not of RUNX3 or CBFβ in GMR-AML1 cells (Fig. 2A). In comparison with the management, exon 5 gRNA greater than exon 4 gRNA, elevated the % of G1 and decreased the % of cells in S and G2/M phases of the cell cycle, probably on account of better decline within the ranges of the cell cycle regulatory oncoproteins (Fig. 2B). Subsequent, the identical variety of GFP optimistic (RUNX1 KO) and detrimental (Ctrl) GMR-AML1 cells have been blended and co-cultured for 30 days. The share of GFP-positive cells was decided through move cytometry, following passage of the cells each 3 days in tradition. On this aggressive in vitro tradition assay, depletion of RUNX1 through RUNX1-KO was related to marked decline within the GFP + GMR-AML1 in comparison with the management cells (Fig. 2C). In comparison with sgRNA management, RUNX1 knockout in GMR-AML1 cells additionally decreased engraftment potential in bone marrow, liver, and spleen of NSG mice throughout an in vivo competitors assay (Fig. 2D and S2A). Moreover, NSG mice engrafted with GMR-AML1 cells during which RUNX1 was knocked out, in comparison with sgRNA control-treated cells engrafted within the mice, exhibited considerably delayed development and extended survival (Fig. 2E).
We had beforehand reported that, in comparison with AML cells expressing two copies of wtRUNX1, within the isogenic AML cells expressing heterozygous mtRUNX1, RUNX1 depletion brought about better sensitivity to homoharringtonine (HHT or omacetaxine)-induced cell dying, related to decreased ranges of c-Myc, c-Myb, MCL1 and Bcl-xL [12]. In current research additionally, publicity to HHT for 48 h dose-dependently induced lack of viability in GMR-AML1 cells, whereas therapy with venetoclax was ineffective (Fig. 2F), probably on account of comparatively excessive expression of MCL1, Bcl-xL and BFL-1 (Fig. 1J and vide infra) [22, 23]. Moreover, therapy with daunorubicin, panobinostat (a pan-HDAC inhibitor), cytarabine or etoposide additionally dose-dependently induced apoptosis in GMR-AML1 cells (Fig. S2B) [24]. Nevertheless, GMR-AML1 cells have been insensitive to A1155463 (Bcl-xL inhibitor), AZD5991 (MCL1 inhibitor), the DNA hypomethylating medication, azacytidine and decitabine, or INCB059872 (LSD1 inhibitor inhibitor)-induced lack of viability (Fig. S2B) [25]. Along with GMR-AML1 cells, we additionally decided the dose dependent-lethal results of HHT on the BMA cells from 11 sufferers with FPD versus 8 sufferers with FPD-MM (Fig. 2G). As proven in Fig. 2G, HHT exerted better deadly exercise towards FPD-MM as in comparison with FPD cells. Fig. S2C, D present the oncoplot of the co-mutations and particular RUNX1 mutations in every of the BMA pattern cells from FPD versus FPD-MM sufferers, respectively.
Results of HHT on chromatin accessibility and gene expressions in GMR-AML and FPD-MM cells
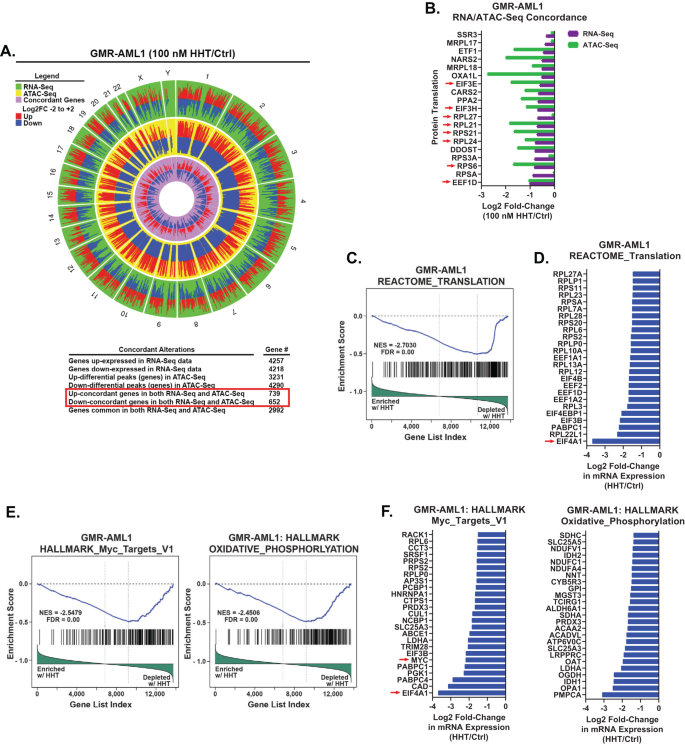
Using bulk RNA-Seq and ATAC-Seq analyses, we in contrast the chromatin accessibility and mRNA gene-expression perturbations in HHT handled (100 nM for 8 h) versus the untreated management GMR-AML1 cells. Genome extensive, 739 genes confirmed vital and concordant improve in ATAC-Seq and RNA-Seq peaks, whereas 652 genes confirmed a concordant decline within the peaks [26], prominently amongst these have been the ribosomal genes concerned in protein translation (Fig. 3A, B). ATAC-Seq and RNA-Seq peaks additionally concordantly declined at quite a few RNA-Pol II transcribed genes, together with DNA POLR2B, TBL1X, CHD4, IGFBP3, CCNE1, CCND2, HDAC5/7, PTPN11, Caspase-8 and RRM2 (Fig. S3). RNA-Seq evaluation of GMR-AML1 cells confirmed that the reactome of gene expressions involving protein translation was negatively enriched, with vital decline within the mRNA of EIF4A1 (Fig. 3C, D). HHT therapy additionally negatively enriched gene expressions within the gene-sets of MYC targets and oxidative phosphorylation in GMR-AML1 cells (Fig. 3E, F).
A, B GMR-AML1 cells have been handled with 100 nM of HHT for eight hours and ATAC-Seq and RNA-Seq analyses have been carried out. A Circos plot and the log2 fold-change of chosen concordant ATAC-Seq and mRNA expression alterations in HHT-treated GMR-AML1 cells are proven. C–F Gene set enrichment evaluation (GSEA) and chosen goal fold modifications in HHT-treated GMR-AML1 cells in contrast with HALLMARK and REACTOME pathway datasets.
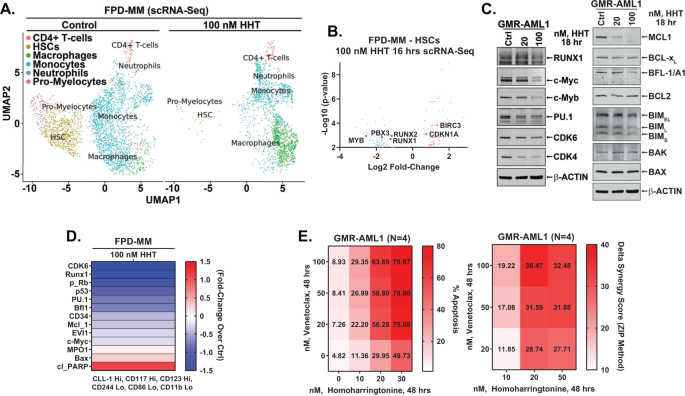
Along with GMR-AML1 cells, using scRNA-Seq analyses, we additionally decided the consequences of HHT therapy on mRNA expressions in PD FPD-MM BMA cells [12, 26]. Figures 4A and S4A show that, following HHT therapy, there was a marked discount within the HSC and pro-myelocyte populations of cells, whereas the cell numbers of macrophages elevated markedly. Within the residual HSC cluster of cells, there was log2-fold decline in MYB, PBX3 and RUNX1/2 mRNA expressions, however vital improve in mRNA of CDKN1A and BIRC3 (Fig. 4B). Bulk RNA-Seq carried out on an FPD-MM pattern additionally confirmed optimistic enrichment of the mRNA expressions belonging to the gene-sets (HALLMARK) of apoptosis signaling and TP53 targets however detrimental enrichment of c-Myc targets (Fig. S4B). Notably, following HHT therapy, there was additionally detrimental enrichment of the reactomes of G2/M checkpoint, DNA replication, cell-cycle checkpoints, E2F mediated regulation of DNA replication, mitotic spindle checkpoint and nuclear envelope breakdown (Fig. S4C). RNA-Seq evaluation of FPD-MM cells additionally confirmed that HHT therapy brought about log2-fold decline in mRNA of MYC, MYB, MCM2/4, FOXM1 and myeloperoxidase (MPO) however improve in CDKN1A, CDKN2B, BCL2A1, ATF3/4, PMAIP1 and HMOX1 (Fig. S4D). Notably, RNA-Seq analysis of a separate PD FPD-MM cell pattern revealed comparable results of HHT therapy on gene-expressions (Fig. S4E–J). We additionally in contrast baseline mRNA expressions and response to HHT in GMR-AML1 and the 2 FDP-MM samples. As proven in Fig. S4K, L, we noticed marked similarity in gene expression at baseline and following HHT therapy within the two FPD-MM samples and GMR-AML1 cells. Following HHT therapy, immunoblot analyses confirmed that, in comparison with untreated controls, GMR-AML1 cells exhibited decreased protein ranges of c-Myb, c-Myc, PU.1, CDK4/6, MCL1, BFL1, Bcl-xL and RUNX1 (Fig. 4C). Mass cytometry carried out on the identical BMA cells confirmed that within the phenotypically characterised FPD-MM stem cells, based mostly on excessive floor expression of CLL1 (CLEC12A), CD117, and CD123, however low expression of CD11b, CD244 and CD86, HHT therapy decreased the expression of CD34, c-Myc, EVI1, MCL1, Bfl1, p53, p-RB, PU.1, RUNX1 and CDK6 (Fig. 4D). The consequences of HHT therapy on protein expressions additionally clarify why, though GMR-AML1 cells have been comparatively insensitive to therapy with venetoclax, cotreatment with HHT and venetoclax synergistically induced apoptosis of GMR-AML1 cells (Fig. 4E).
A Major FPD-MM18 cells have been handled with 100 nM of HHT for 16 h and single cell RNA-Seq (scRNA-Seq) evaluation was carried out. SingleR evaluation was utilized to outline cell clusters. The UMAP plot exhibits outlined cell populations. B Log2 fold-change in mRNA expressions on account of HHT therapy within the HSC inhabitants. C GMR-AML1 cells have been handled with the indicated concentrations of HHT for 18 h. Following this, immunoblot analyses have been carried out on complete cell lysates. The expression of β-actin within the lysates served because the loading management. D Affected person-derived FPD-MM18 cells have been handled with 100 nM of HHT for 18 h. Cells have been harvested and analyzed by CyTOF evaluation utilized a cocktail of uncommon steel element-tagged antibodies. Leukemia stem cells have been outlined by excessive expression of CLEC12A(CLL-1), CD117, and CD123 and low expression of CD244, CD86, and CD11b. Warmth map exhibits absolutely the fold-change of considerably altered protein expressions within the handled over management cells. E GMR-AML1 cells have been handled with the indicated concentrations of HHT and/or venetoclax for 48 h. Then the proportion annexin V-positive apoptotic cells was decided by move cytometry. Delta synergy scores (ZIP) have been decided utilizing SynergyFinder v3.0 internet utility.
Deadly exercise of mebendazole (MB), different anti-mitosis brokers and established anti-AML brokers in GMR-AML1 cells
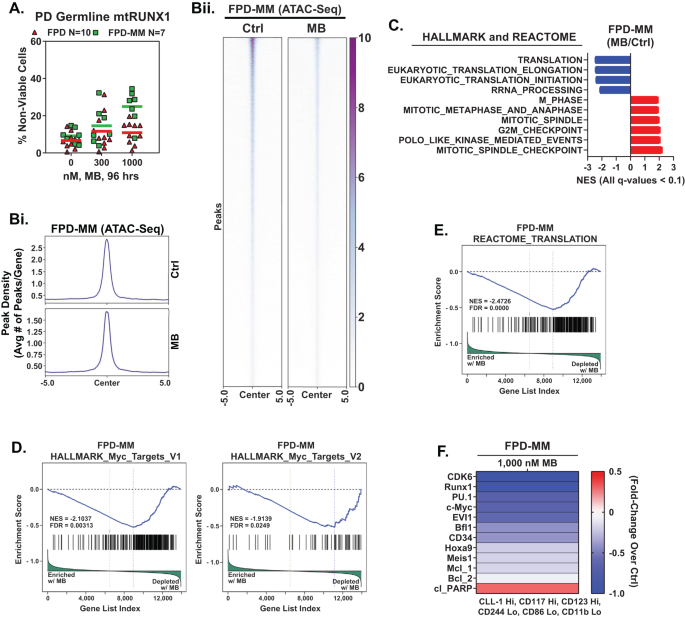
Primarily based on the EMs revealed by the LINCS1000 CMap evaluation described in a earlier report [4], we additionally decided the consequences of mebendazole (MB) on freshly procured FPD-MM vs RUNX1-FPD cells. As proven in Fig. 5A, MB induced better lack of viability in FPD-MM as in comparison with RUNX1-FPD cells. In distinction, MB therapy was markedly much less poisonous towards regular CD34+ HPCs (Fig. S5). We subsequent decided the consequences of MB therapy on chromatin accessibility and transcription in FPD-MM cells. Determine 5B demonstrates that publicity to MB considerably decreased genome extensive chromatin accessibility, as decided by ATAC-Seq peaks density in FPD-MM cells. Following MB therapy of FPD-MM cells, RNA-Seq evaluation revealed vital optimistic enrichment of the gene-sets concerned in G2/M checkpoint, mitotic metaphase and anaphase, mitosis, spindle checkpoint, mitotic spindle and polo-like kinase mediated occasions (Fig. 5C). In distinction, there was additionally detrimental enrichment of the gene-sets of ribosomal RNA processing, eukaryotic translation initiation/elongation and protein translation (Fig. 5C). Particularly, HALLMARK gene-sets of c-Myc targets and the reactome of protein translation have been negatively enriched (Fig. 5D, E). Guided by these MB-mediated mRNA perturbations, we carried out CyTOF evaluation to find out the consequences of MB therapy on particular protein expressions. As proven in Fig. 5F, MB therapy decreased the protein ranges of RUNX1, CDK6, PU.1, c-Myc, EVI1, HOXA9, MEIS1, BFL1 and MCL1 in phenotypically-defined FPD-MM myeloid stem-progenitor cells [26, 27]. In GMR-AML1 cells, according to tubulin polymerization exercise of MB, proven in Fig. 6A, MB therapy for twenty-four h induced improve in % of cells within the G2/M section of the cell-cycle (Fig. 6B). Publicity to >300 nM of MB for 72 h additionally induced lack of viability in GMR-AML1 cells (Fig. 6C). RNA-Seq evaluation carried out in GMR-AML1 cells following therapy with MB revealed detrimental enrichment of the reactome of ribosomal RNA processing, eukaryotic translation initiation/elongation and protein translation, in addition to detrimental enrichment of the MYC targets (Figs. 6D, E, S6A, B). A log2-fold vital decline within the mRNA of ribosomal proteins was noticed within the reactome of protein translation in GMR-AML1 cells (Fig. 6F). RPPA (reversed section protein array) evaluation of GMR-AML1 cells revealed that MB therapy elevated protein expression of p-Aurora A/B/C kinases, Aurora A/B kinase and PLK1, however decreased protein ranges of p-MEK, eEF2K, PI3K-p85, p-eIF4E and MAPK (Fig. 6G). Primarily based on these perturbations on account of MB therapy, we additionally decided the consequences of therapy with MB, volasertib (PLK1 inhibitor) or alisertib (Aurora A kinase inhibitor) on nascent polypeptide elongation [12, 28]. Publicity to MB or volasertib, however not alisertib, considerably inhibited nascent polypeptide elongation in GMR-AML1 cells (Fig. 6H). Certainly, co-treatment with MB and volasertib or plogosertib induced synergistic lethality in GMR-AML1 cells, with delta synergy scores of >10 by the ZIP technique (Figs. 6I and S6C). In distinction, therapy with volasertib didn’t induce vital lethality, or its co-treatment with MB didn’t considerably improve MB-induced lethality in CD34+ regular progenitor cells (Fig. S6D, E). These findings spotlight the deadly exercise and related molecular perturbations on account of MB and/or volasertib towards mobile fashions of FPD-MM.
A PD germline mutant Runx1 FPD and FPD-MM cells have been handled ex vivo with the indicated concentrations of MB for 96 h. Then, the proportion non-viable cells was decided by TOPRO-3 iodide staining and move cytometry. B World ATAC-Seq warmth map and peak profile in PD, FPD-MM17 cells handled with 1000 nM MB for twenty-four h at peak +/− 5 kb decision. C–E Major FPD-MM18 cells have been handled with 1000 nM of MB for twenty-four h and bulk RNA-Seq evaluation was carried out. Gene set enrichment evaluation (GSEA) in MB-treated FPD-MM18 cells in contrast with HALLMARK and REACTOME pathway datasets. F Affected person-derived FPD-MM18 cells have been handled with 1000 nM of MB for twenty-four h. Cells have been harvested and analyzed by CyTOF evaluation utilized a cocktail of uncommon steel element-tagged antibodies. Leukemia stem cells have been outlined by excessive expression of CLL-1, CD117, and CD123 and low expression of CD244, CD86, and CD11b. Warmth map exhibits absolutely the fold-change of considerably altered protein expressions within the handled over management cells.
A GMR-AML1 cells have been handled with the indicated focus of MB for 16 h. Cells have been fastened, permeabilized, and stained with β-Tubulin antibody. DNA have been stained with DAPI. Cells have been imaged by confocal microscopy. Authentic magnification is 60×. B GMR-AML1 cells have been handled with the indicated focus of MB for twenty-four h. Cells have been fastened with 70% molecular grade ethanol, stained with propidium iodide, and cell cycle evaluation was decided by move cytometry. C GMR-AML1 cells have been handled with the indicated concentrations of MB for 72 h. Then the proportion apoptotic cells have been decided by Annexin V and TO-PRO-3 iodide staining and move cytometry. D–F GMR-AML1 cells have been handled with 1000 nM of MB for 16 h and bulk RNA-Seq evaluation was carried out. Gene set enrichment evaluation (GSEA) in MB handled cells in contrast with HALLMARK and REACTOME pathway datasets. G GMR-AML1 cells have been handled with 3000 nM MB for 16 h in biologic triplicates. Reverse section protein array (RPPA) evaluation was carried out. Log2 fold-changes in chosen considerably altered proteins are proven. H GMR-AML1 cells have been handled with the indicated concentrations of MB, Volasertib, or Alisertib for 16 h. Nascent polypeptide elongation was detected by OPP puromycin incorporation assay and move cytometry. Column represents imply of six unbiased experiments; bar represents SEM. I GMR-AML1 cells have been handled with the indicated concentrations of MB and/or Volasertib for 48 h. Then the proportion annexin V-positive apoptotic cells have been decided by move cytometry. Delta synergy scores (by ZIP technique) have been decided utilizing SynergyFinder v3.0.
In vivo efficacy of omacetaxine (HHT), MB and/or volasertib in a xenograft mannequin of GMR-AML1 cells
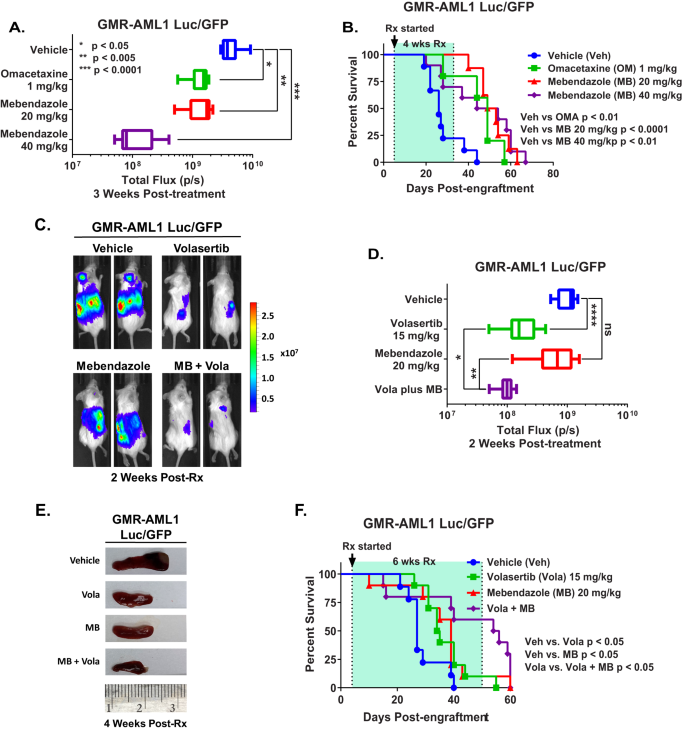
We subsequent decided the in vivo anti-leukemia efficacy of omacetaxine (HHT or OM) or mebendazole within the xenograft mannequin of luciferase/GFP transduced GMR-AML1 cells. Following tail vein infusion and engraftment of GMR-AML1-Luc/GFP cells, cohorts of NSG mice have been handled with car management or beforehand decided protected doses of the medication [12, 14]. Determine 7A demonstrates that following three weeks of therapy, single agent OM or MB, in comparison with car management, considerably decreased leukemia burden on account of GMR-AML1 cells, with better discount brought on by therapy with 40 mg/kg of MB. Following 4-weeks of therapy, every drug in comparison with car management additionally considerably improved survival of NSG mice, with out inducing toxicity (Fig. 7B). In a separate examine, cohorts of mice engrafted with GMR-AML1-Luc/GFP cells have been handled with car management, MB and/or volasertib. In comparison with car management, though therapy with volasertib considerably decreased leukemia burden and spleen dimension, co-treatment with volasertib and MB was considerably much more efficient in lowering the leukemia burden in addition to in lowering the spleen dimension in NSG mice (Fig. 7C–E). Following 6 weeks of therapy, in comparison with car management, therapy with volasertib or MB considerably improved survival of the NSG mice. Furthermore, co-treatment with MB and volasertib was considerably simpler than volasertib alone in bettering survival of NSG mice, once more with out inducing toxicity (Fig. 7F). These findings spotlight the one agent in vivo efficacy of OM, MB and volasertib, in addition to superior efficacy of co-treatment with MB and volasertib towards the xenograft mannequin of GMR-AML1 cells.
A Whole bioluminescent flux (P/S) in NSG mice engrafted with (1 million) GMR-AML1 cells and handled for 3 weeks as indicated. B Kaplan–Meier survival curve of NSG mice engrafted with (1 million) GMR-AML1 cells and handled for 4 weeks as indicated. Significance was decided by a Mantel-Cox log rank take a look at. C Consultant pictures of mice engrafted with GMR-AML1 Luc/GFP cells and handled with 20 mg/kg MB and/or 15 mg/kg Volasertib for 2 weeks and IVIS imaged. D Whole bioluminescent flux (P/S) in NSG mice engrafted with (1 million) GMR-AML1 cells and handled for 2 weeks as indicated. *p < 0.05; **p < 0.005; ****p < 0.0001. E Consultant pictures of spleens from mice engrafted with GMR-AML1 Luc/GFP cells and handled with 20 mg/kg MB and/or 15 mg/kg Volasertib for 4 weeks. F Kaplan-Meier survival curve of NSG mice engrafted with (1 million) GMR-AML1 cells and handled for six weeks as indicated. Significance was decided by a Mantel-Cox log rank take a look at.